This tweet wondered about a 'decaf pill' to add to drinks. I though about proteins or something that would bind (but you'd need 25g or something), enzymes to break it down (but you'd need to work in hot + acidic conditions and be food safe). But e.g. activated charcoal binds caffeiene, would that work? Test:
- Two identical ~80ml portions of instant decaf, with 100mg pure caffeine added (10ml @ 10mg/ml).
- Coffee filter with activated charcoal bed prepared, briefly washed (not enough, it turns out)
- One coffee passed through this twice then filtered through a clean filter to get rid of at least some of the charcoal dust
- re-heated to same temp, small cups used for blind taste test
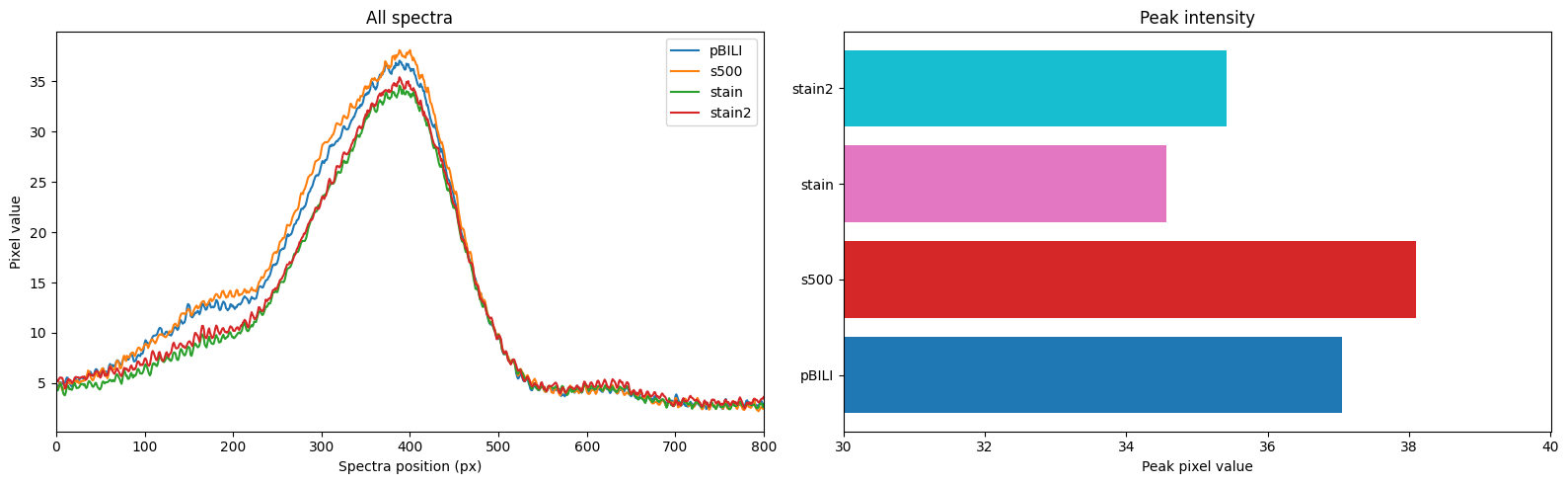
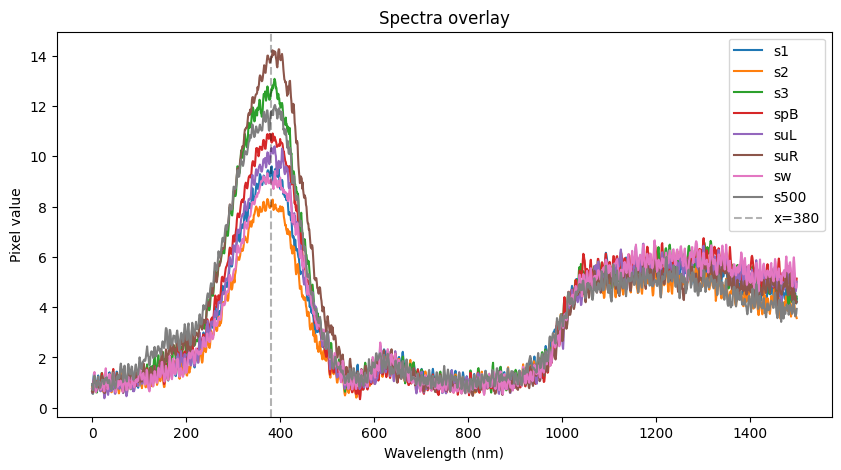
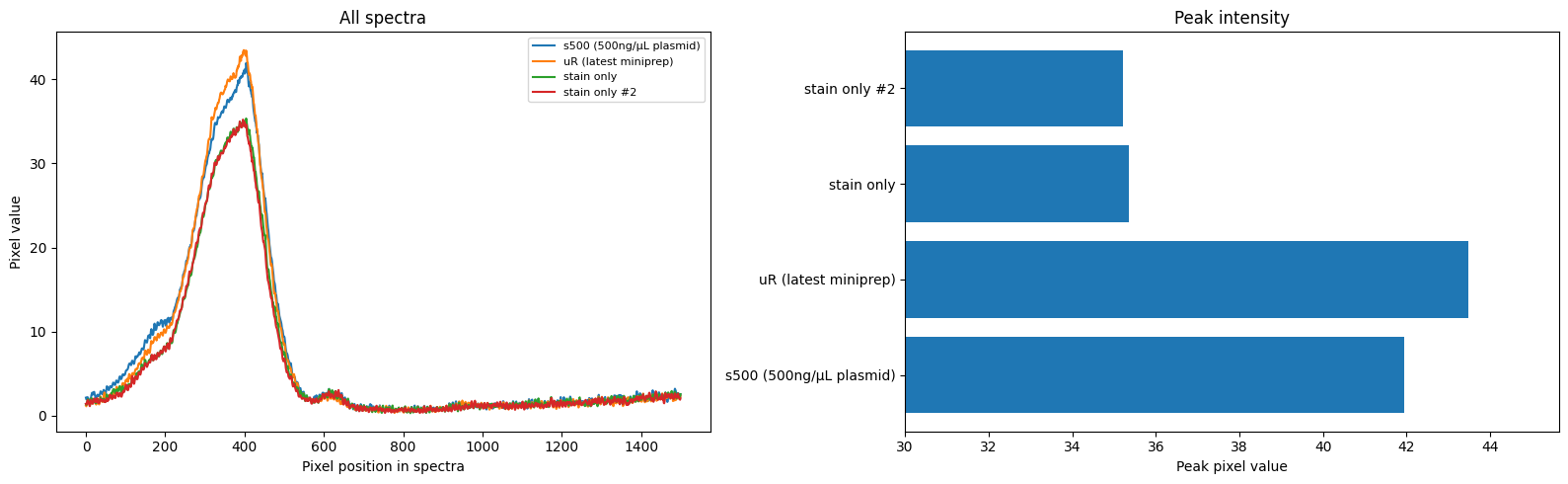
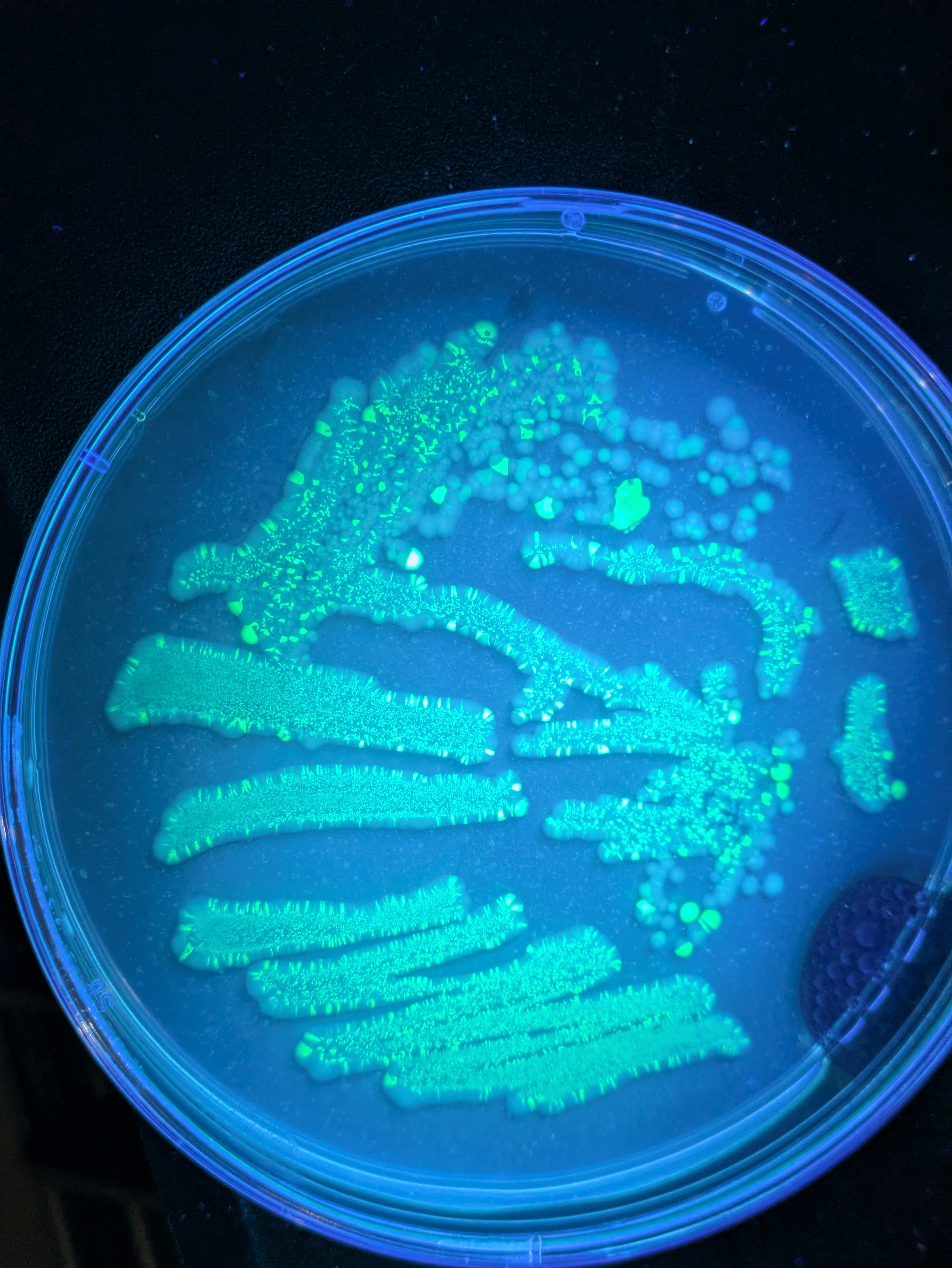
- 7uL spot on TLC plate of each, run with ethyl acetate, compared to known standards with 254nm illumination
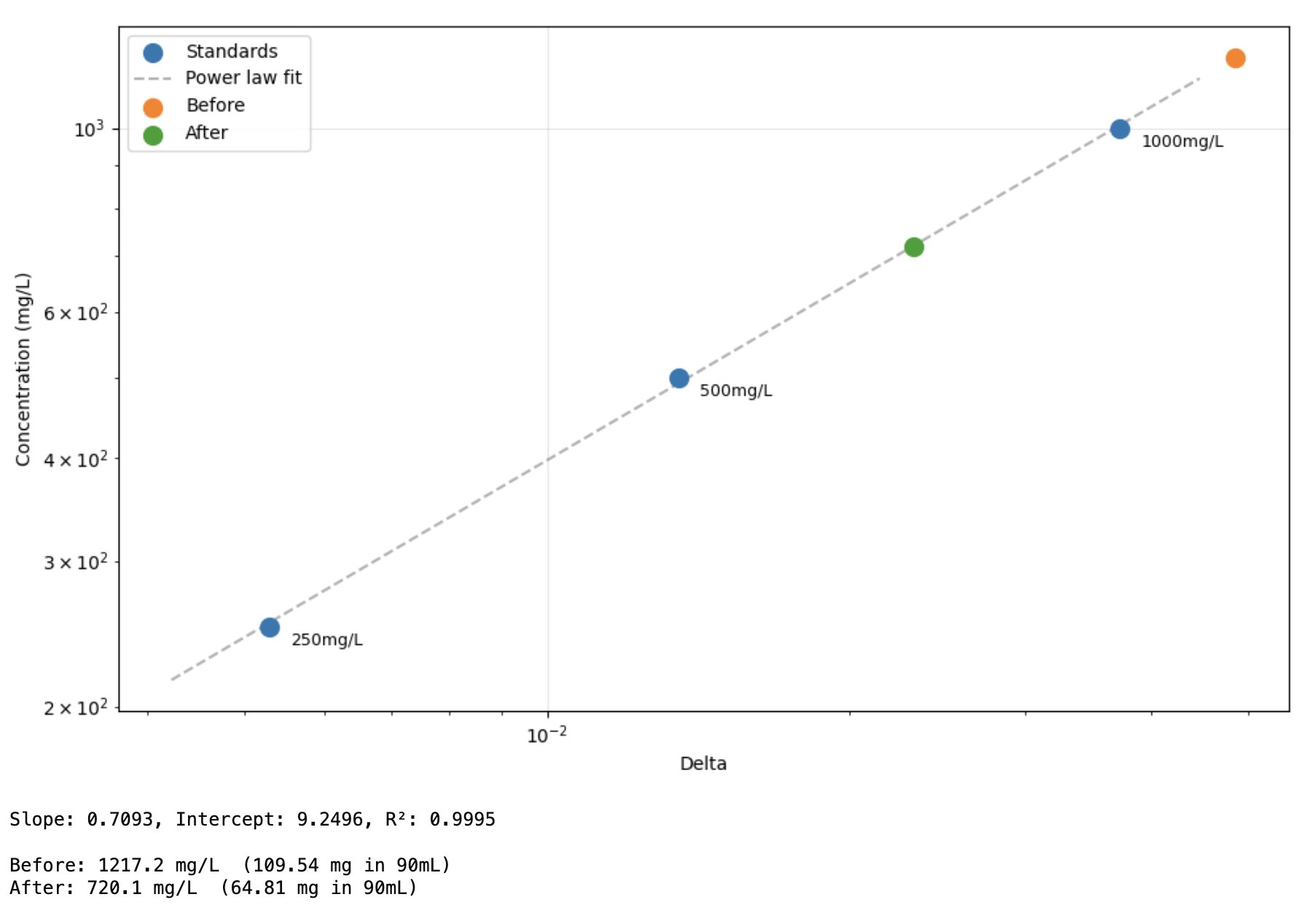
Results: TLC shows a significant reduction in caffeine, but also less polyphenols etc (judging by glow under 356nm UV torch). I found the non-filtered reference unpleasant and bitter, I actually guessed it was the filtered version and something had gone wrong! The filtered 'decaf' version tasted smooth but bland, I preferred it although this is an unrealistic caffeine concentration for coffee and there was no milk etc. (edit: added milk, closer to a tie (both bad)).
I bet you could end up with some kind of tea bag form factor with better washed activated charcoal, that you add to coffee or whatever for a while before drinking. Maybe good in a pinch if the only options are caffeinated and you don't mind a bit of flavour change...
Edit: fitting a quick curve it looks like only 50% reduction, less than I guessed by eyeballing (although likely about as accurate tbh)