Tuesday, March 3, 2026
Duckweed tfm
PermalinkNotebook Page
Experiment Gallery




Daily Notes
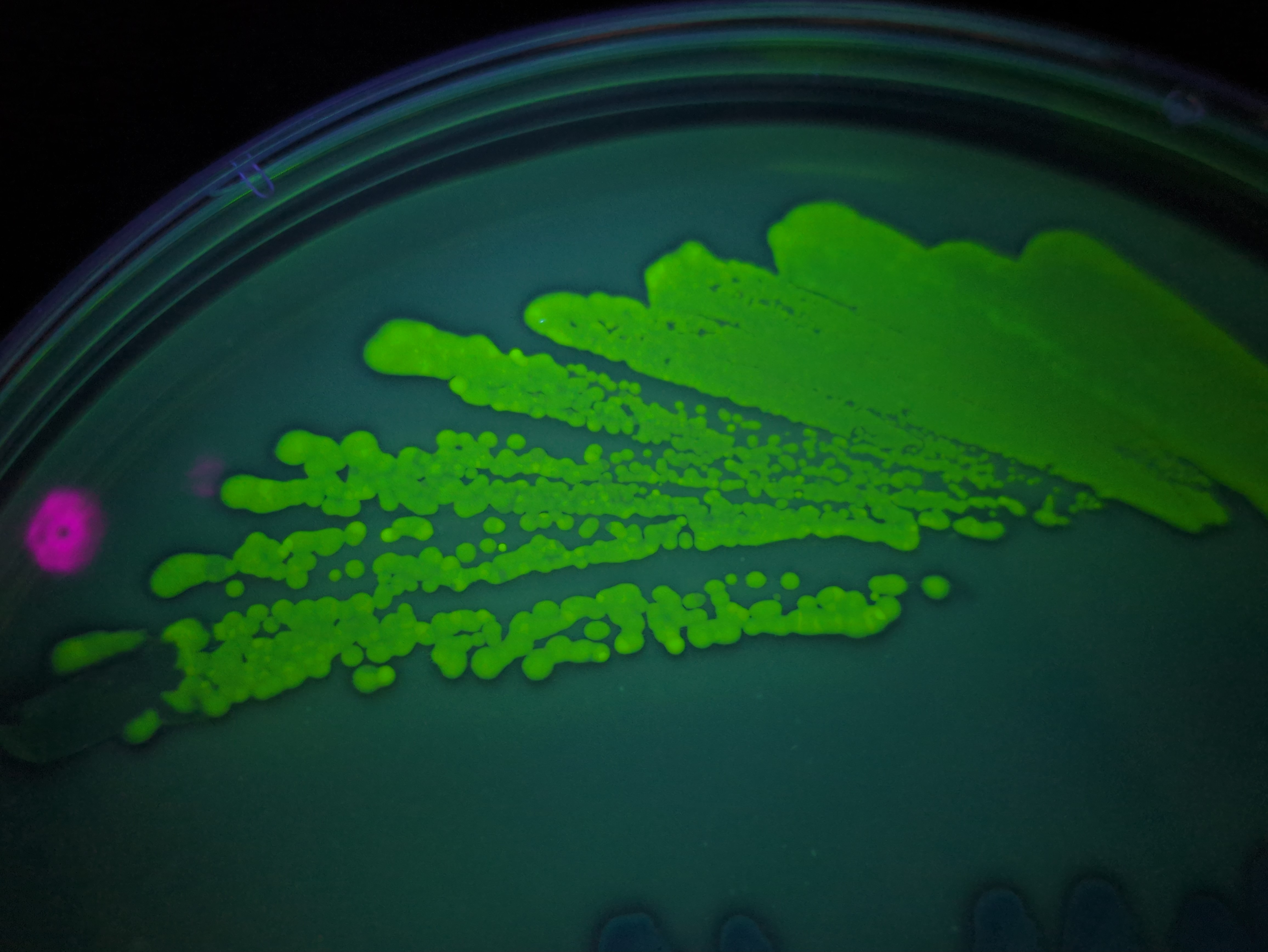
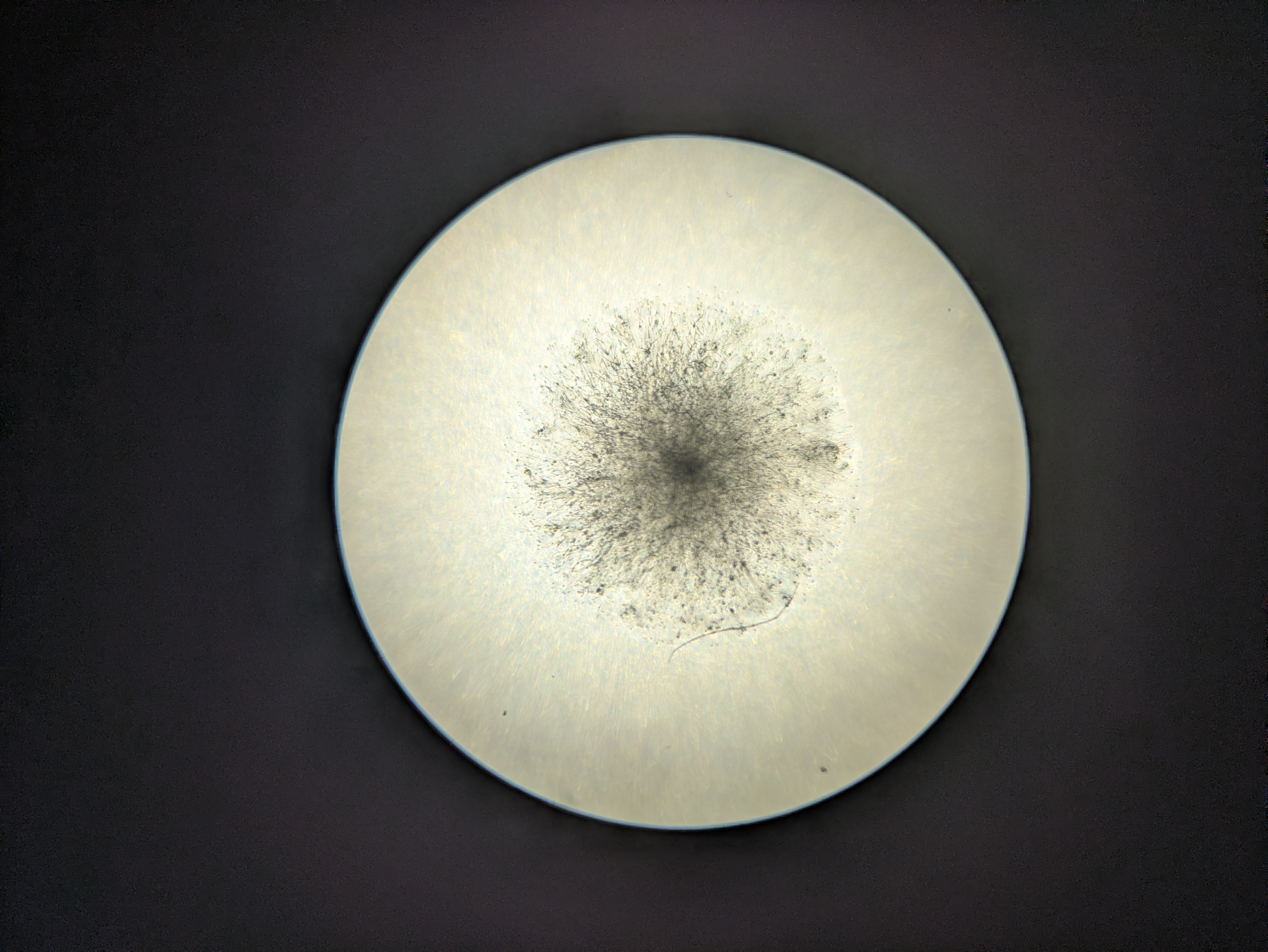
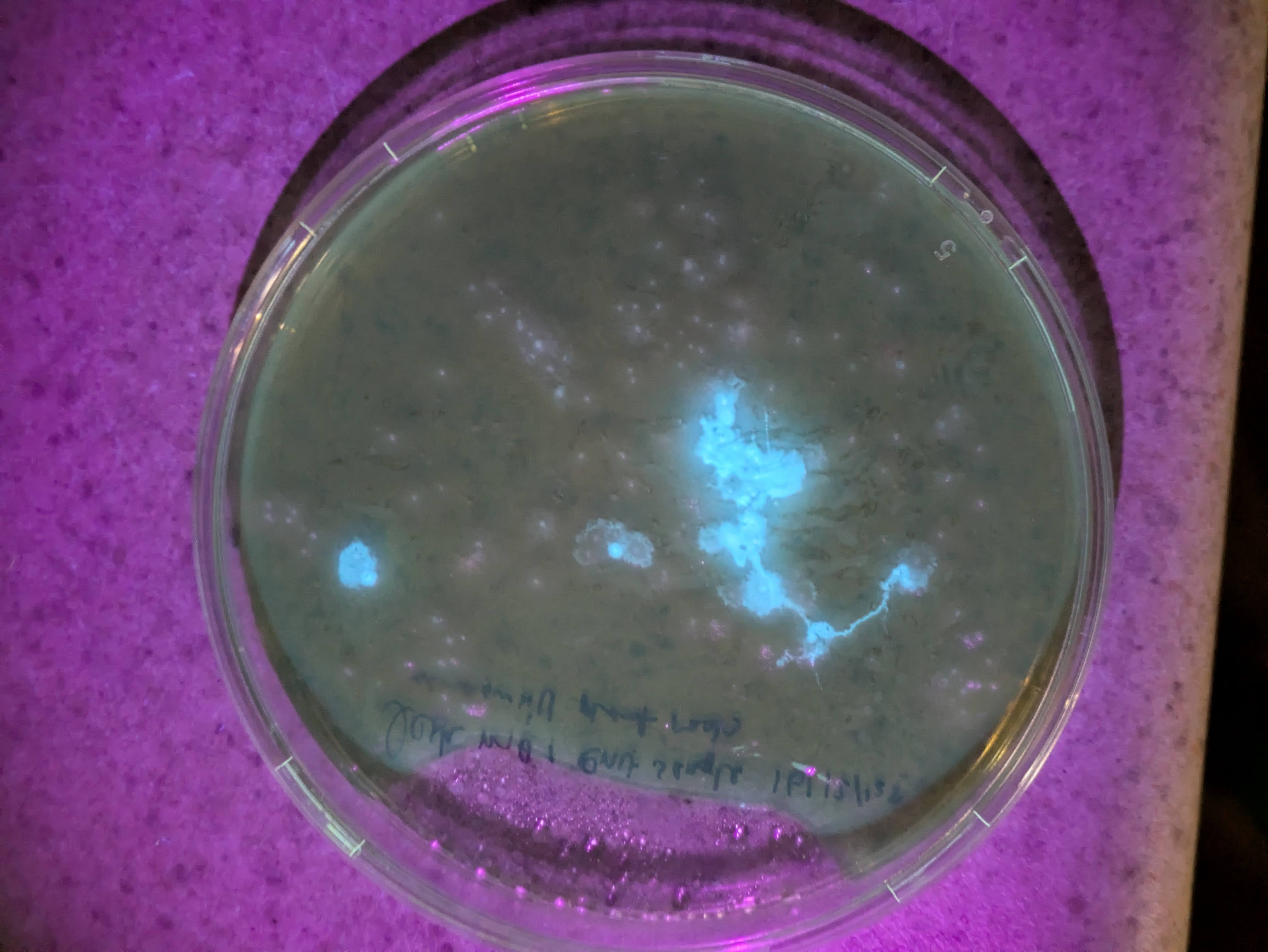
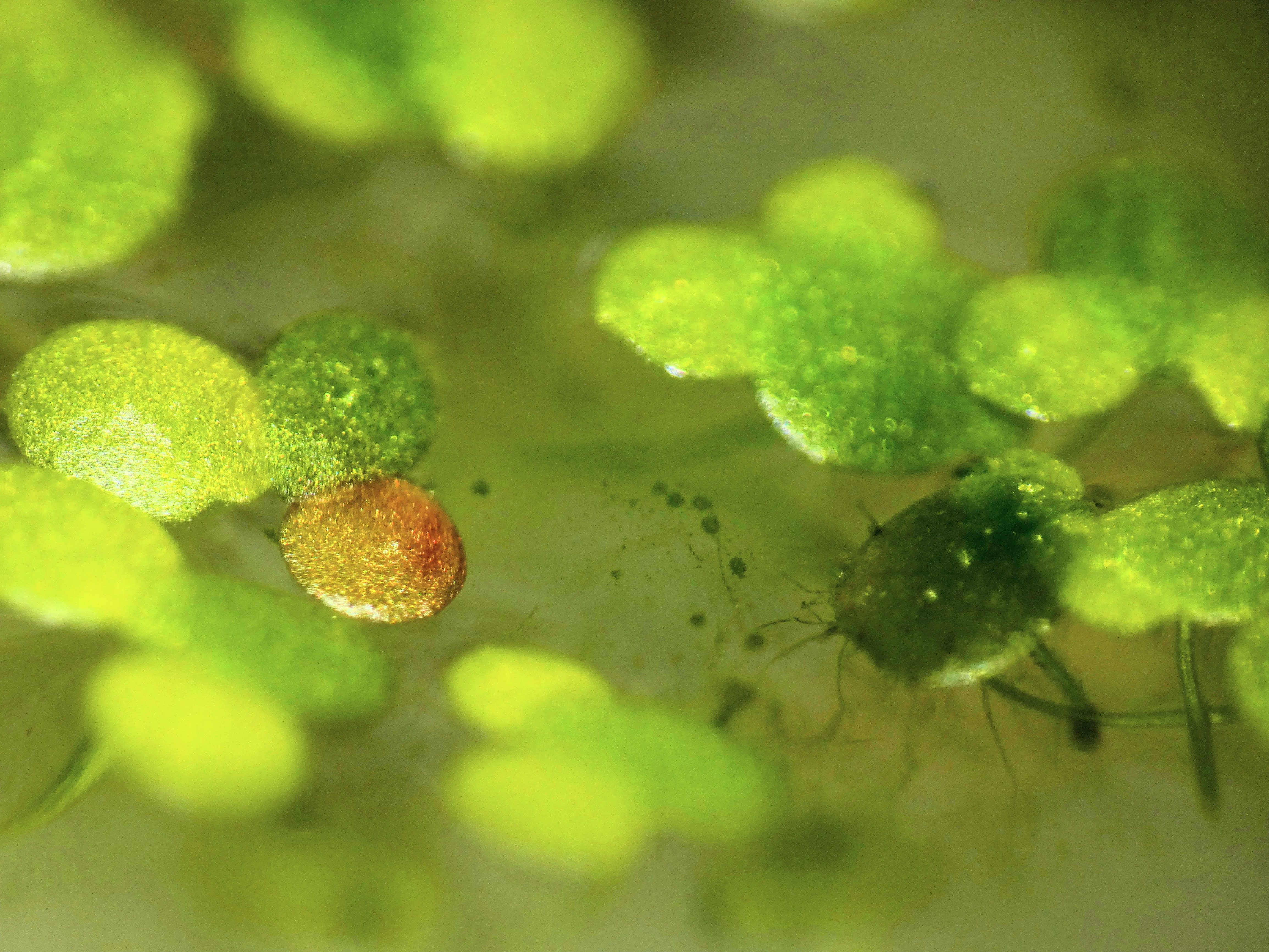
Attempting tissue-culture-free agro transform of duckweed (lemna minor). I ran a few diff exps this weekend, and more excitingly noticed a red frond in some that I'd tried to tfm on Feb 9th, hoping that means the meristem took in the genes and this is a properly transgenic new frond. Rough protocol:
- A loopful of agro into infiltration mix, in a syringe with some duckweed fronds
- Pull back plunger while blocking end, and release. The sudden vacuum+slam (in my head at least) helps push agro into the interstices of the fronds.
Will share more depending on how more recent tests go.