Notebook Page
Experiment Gallery


Daily Notes
I made some liquid cultures yesterday, and today am trying a wash step before electroporation. First test in the am:
- 1.5ml pp culture, spin down, remove supernatant
- Add 1.5ml water, spin down, remove ~almost all of it
- Resuspend pellet, ~30uL liquid total, and place on 1.2mm gap
- Add 5uL DNA (@10ng/ul)
- Zap 5-10 times
- Into 100uL LB broth, warm (~30C) for an hour, plate.
I like P. putida - it got cloudy much faster than e.c. (first pic shows after ~12h maybe?) and doesn't smell like poop. Will run more tests later today / tomorrow as time permits, I might also want to switch to DNA that has a visile marker, I ended up with single colonies on a few plates last time and I'm not sure if they are just contam or survivors that evolved resistance or if they actually took in the plasmid, maybe I'll mini-prep one of sebs chromoprotein colors so I have plenty of e.g. GFP plasmid for testing this. Nice Saturday project option.
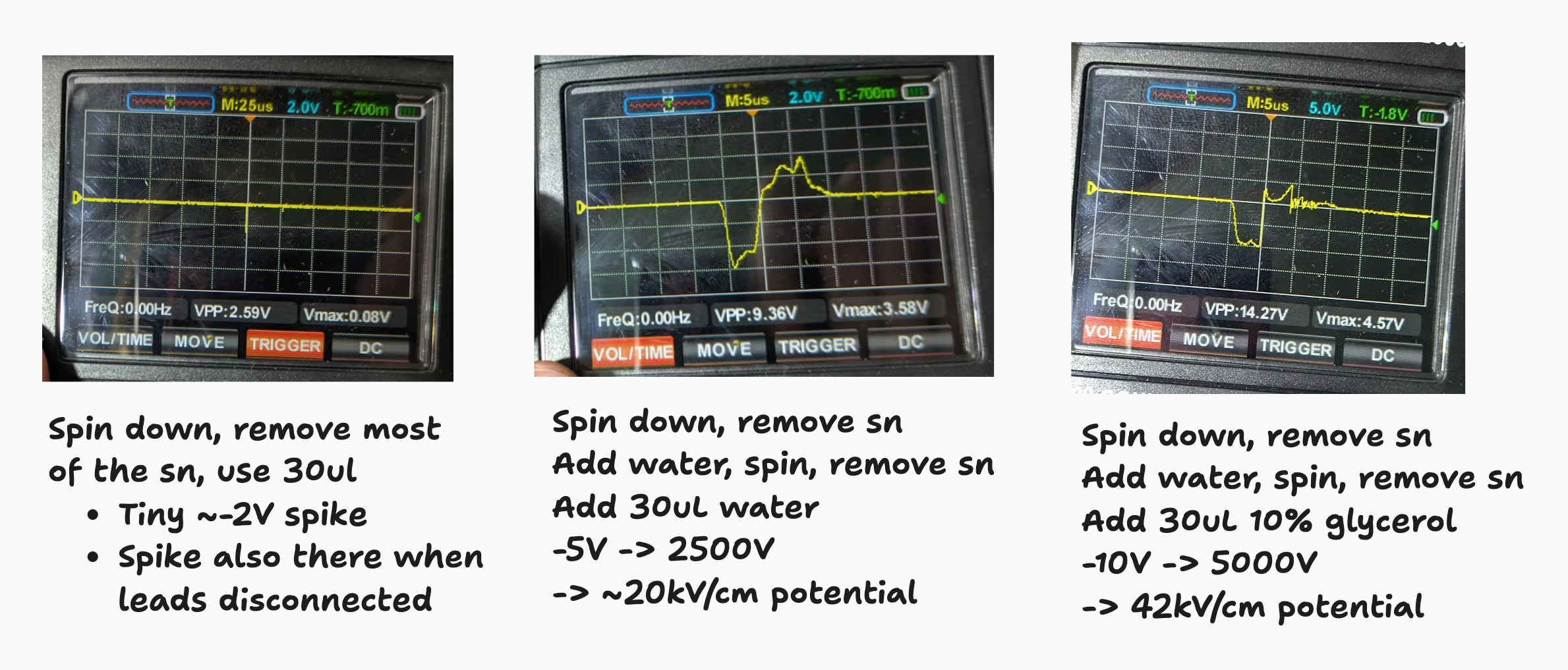
Later: Hooked up my oscilloscope with a 10k resistor in series with a 5M to cut down the voltage, then ran it with a few configurations:
Open: -11V spike
Cells in liquid culture spun down once: sometimes a very short spike of a 1-2V
Cells in LC, spun down, resuspended in water, spun down, then in 30uL water: -5V for 5us (then a smaller +ve peak too)
Cells in LC, spun down, resuspended in water, spun down, ~all the supernatant removed, then suspended in 30uL 10% glycerol: -10V spike for 5us (then a smaller +ve peak too)
All this with a 1.2mm gap. Pinch of salt: get a spike of a few V even with leads disconnected, and 480k is a big load for a piezo. Still - clearly the washing is actually needed, I should probably do several washes next time I try this properly.