Friday, March 27, 2026
Transformed Duckweed
PermalinkExperiment Gallery






Daily Notes
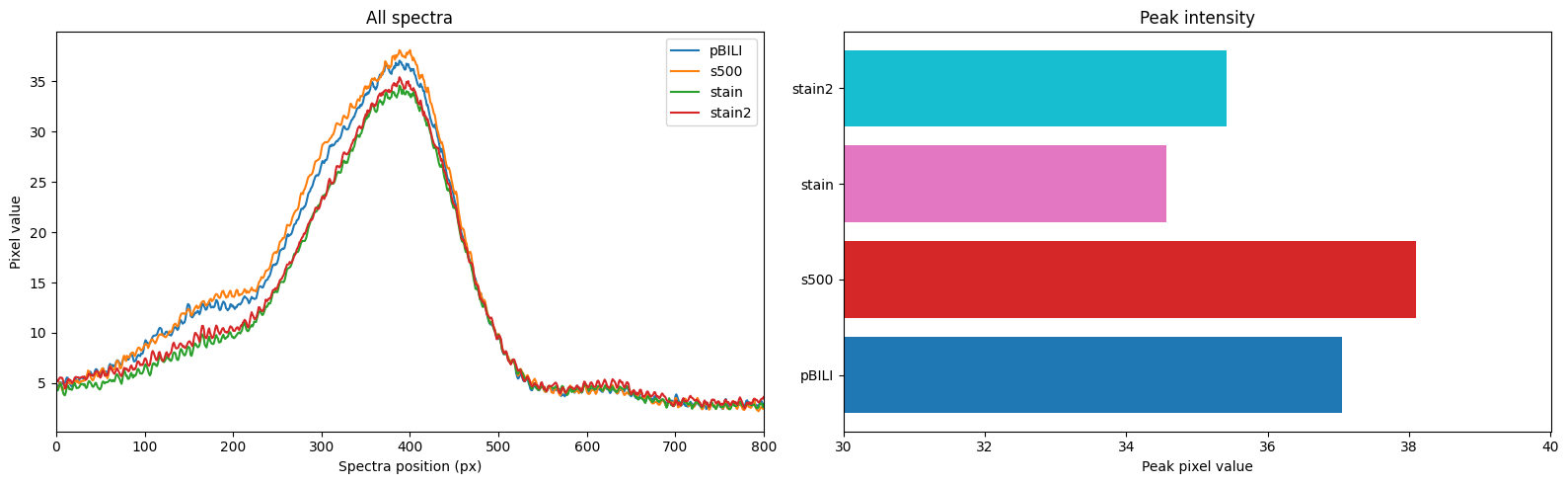
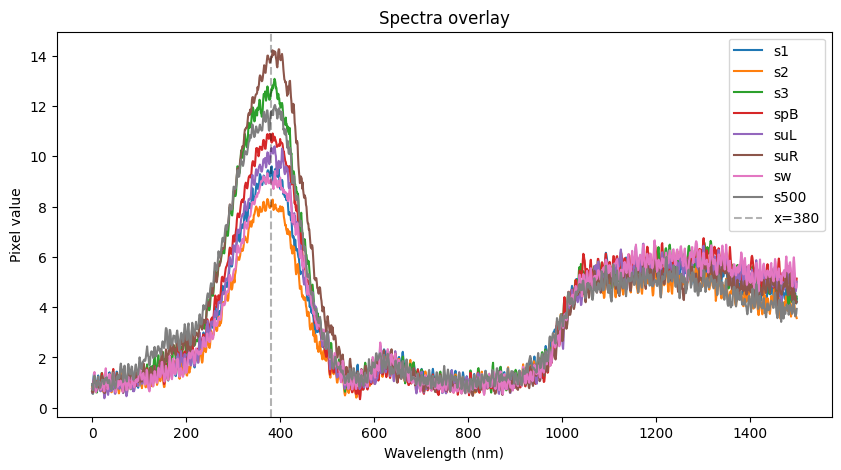
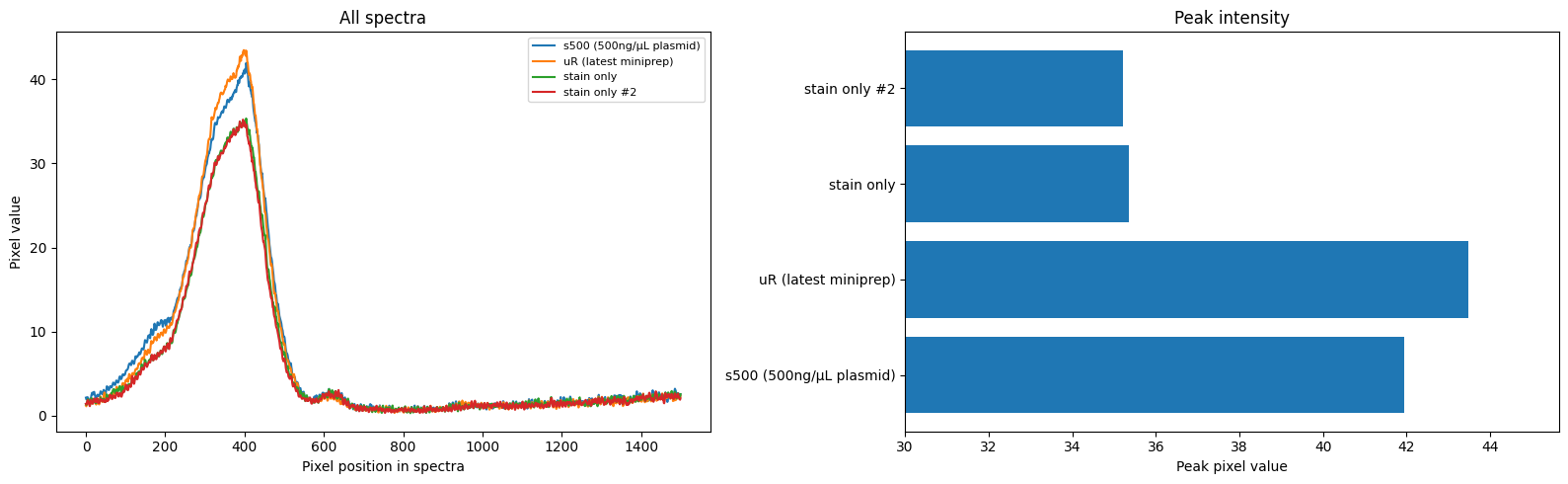
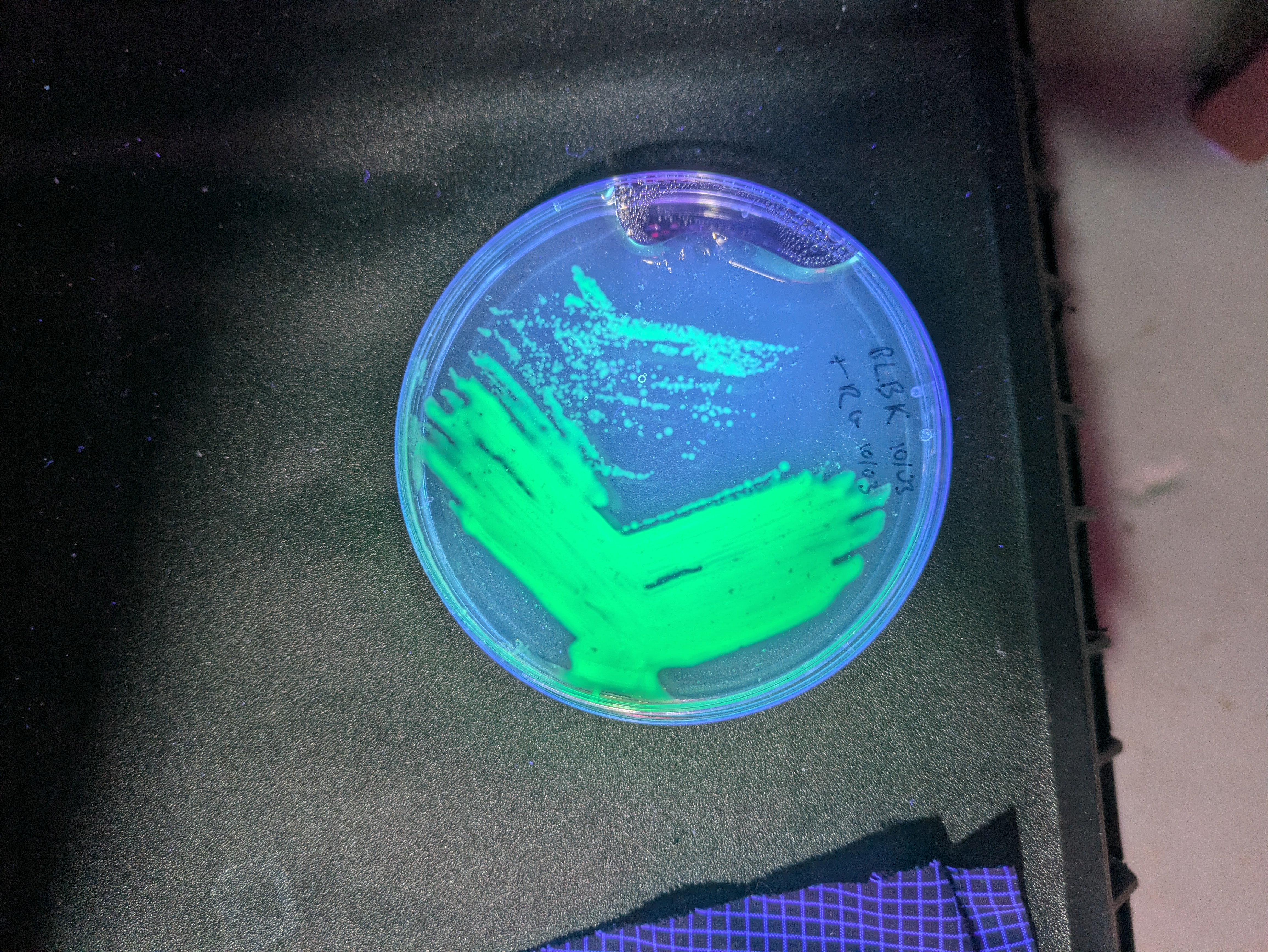
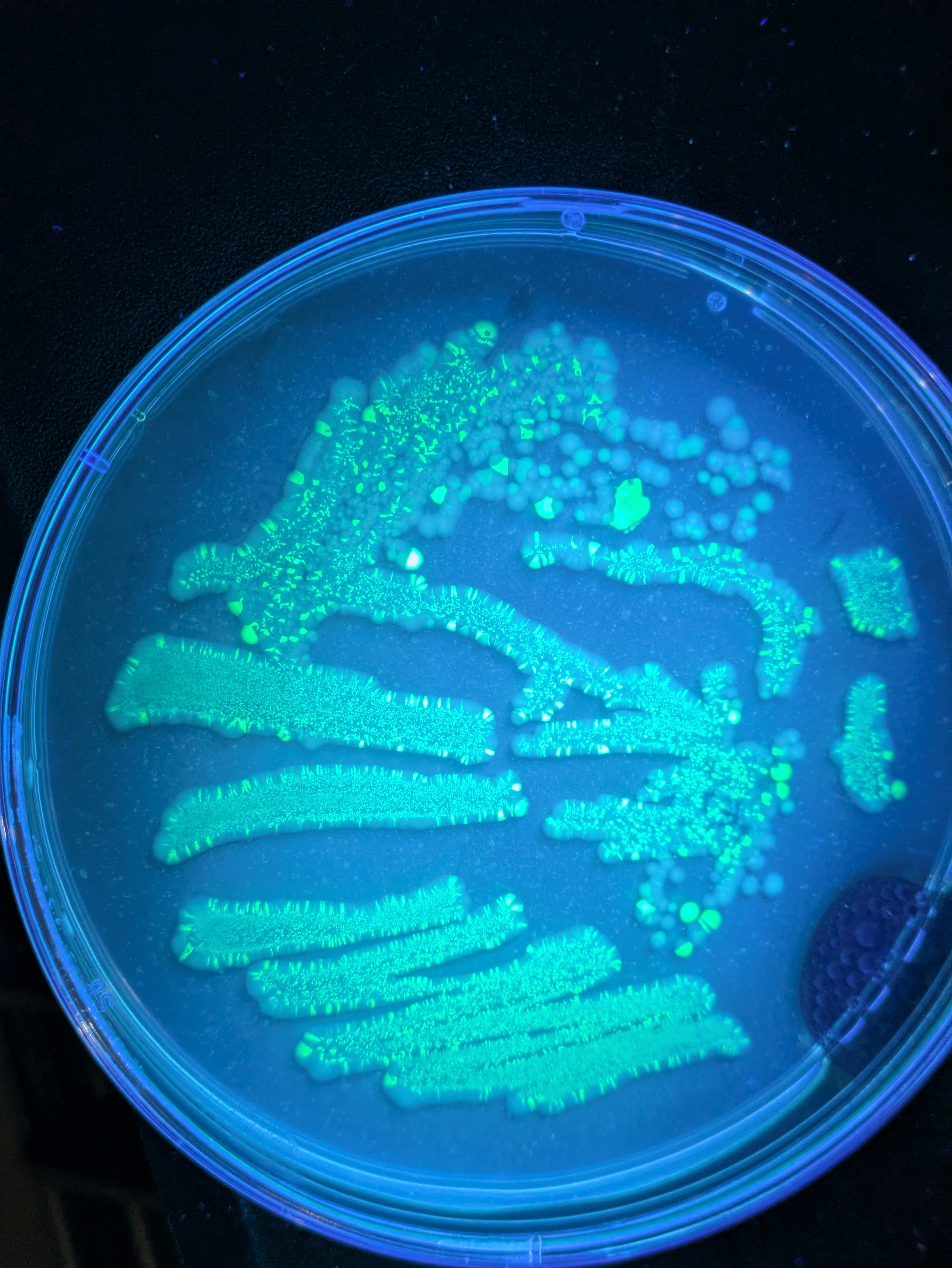
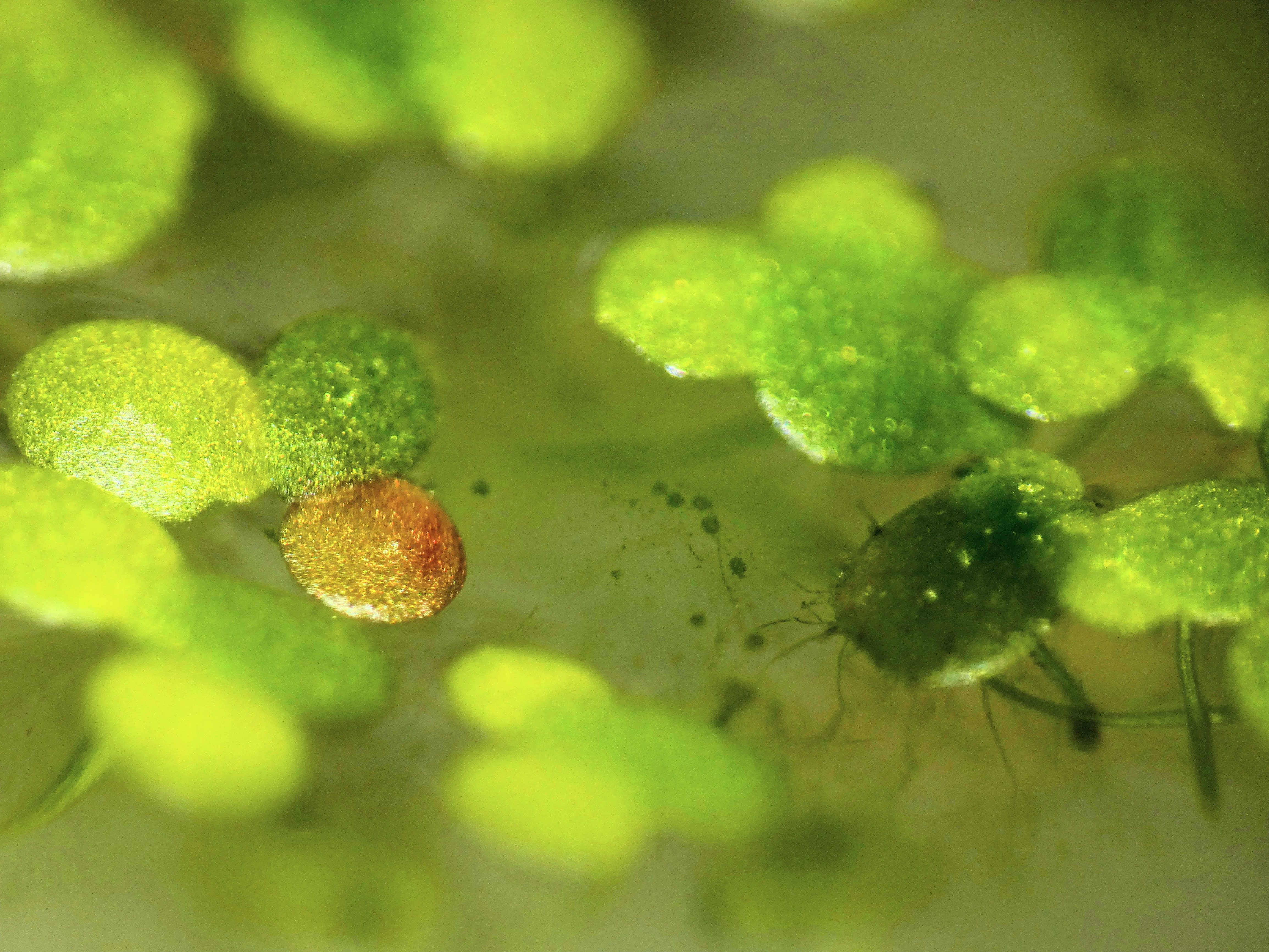
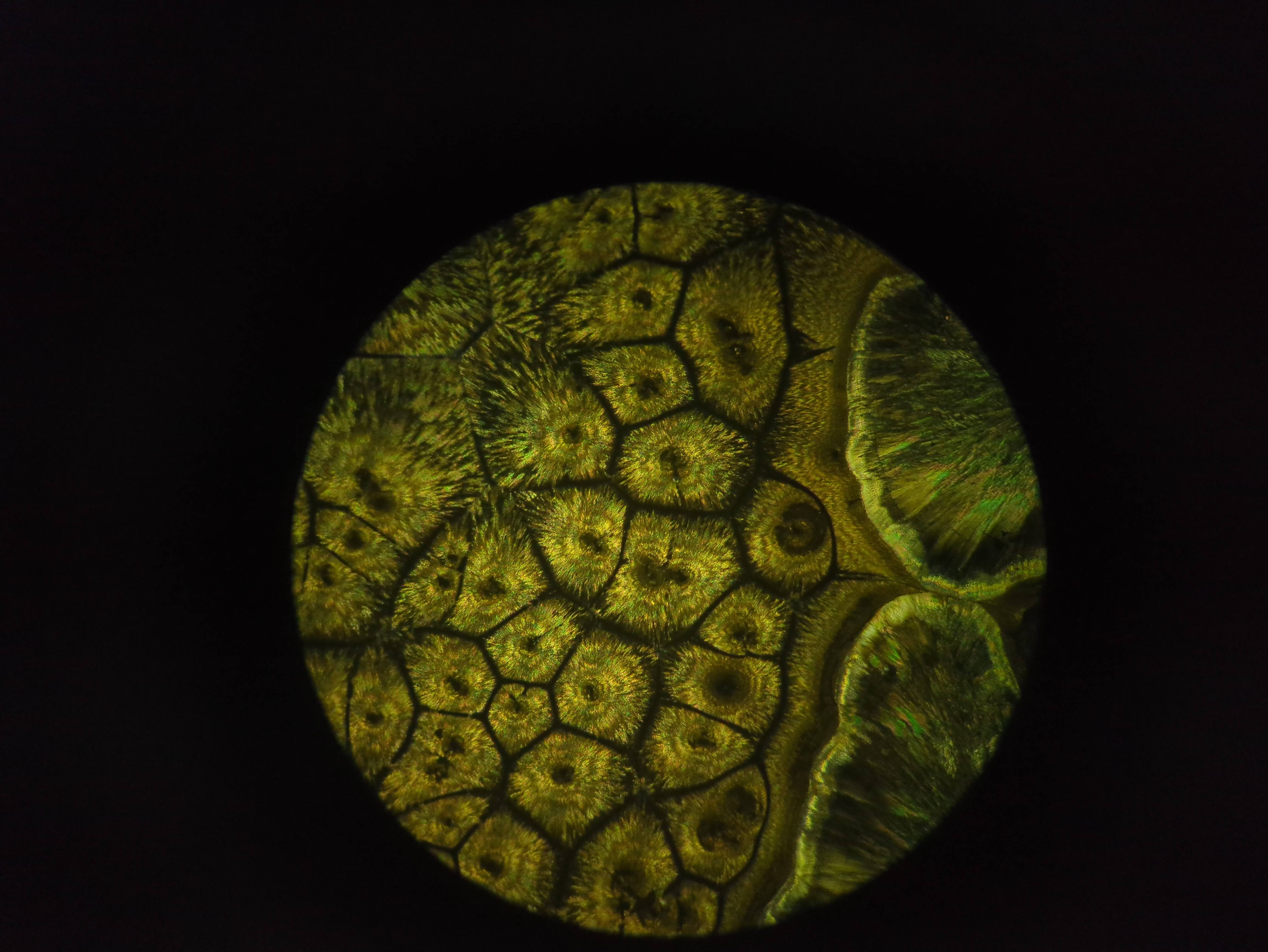
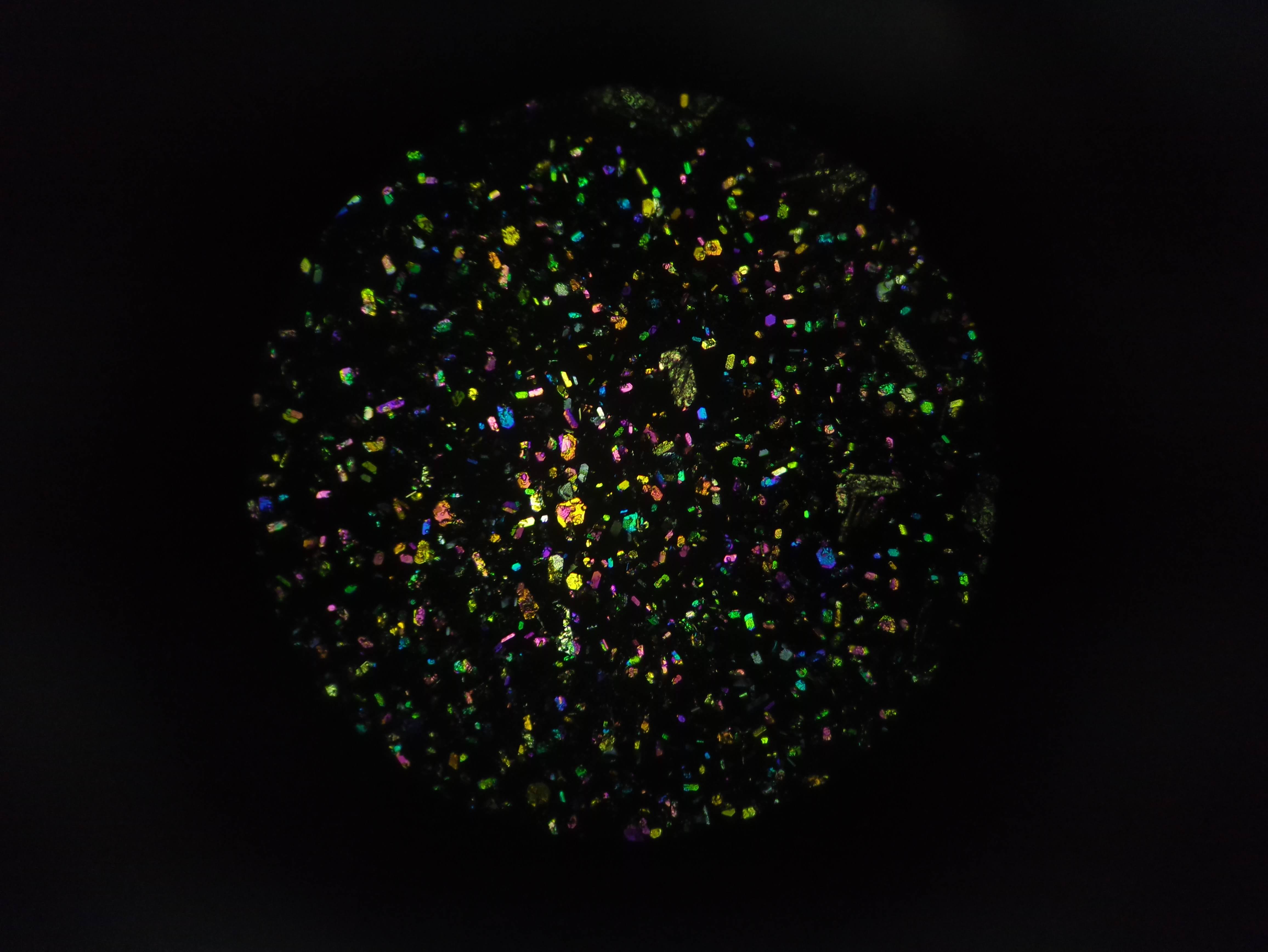
More pics of duckweed with RUBY, transformed with agrobacterium. I still need to try a fresh attempt, planning to split them in hopes of transforming more meristems, but even the rough and ready way I did these at least yields some partial transforms and a couple of fully transformed bits! Hopefully over successive generations we can weed out the mosaic mixes and get pure, but if I can up the initial transformation efficiency that'll help. How these were done:
- Agro + tfm mix in a syringe
- Add some dwe fronds
- Pull back and release plunger a few times
- Rinse a few times
- Into nutrient mix to recover + grow
- Wait.